
Contact Us

Schizophrenia is a complex mental health condition that affects how the brain functions, making it difficult for individuals to think, manage emotions, and engage in daily activities. With advancements in mental health care, TMS therapy for schizophrenia at Neurotherapeutix in New York offers a promising, non-invasive approach to managing symptoms and improving quality of life.
If you or a loved one is living with schizophrenia, our leading-edge treatments can provide effective, personalized care. Contact us today or read on to learn more about our approach.
Schizophrenia is a mental health condition that arises from a combination of genetic and environmental factors.
Studies have identified several risk genes associated with the condition, while environmental influences such as birth complications, prenatal infections, or drug use can increase the likelihood of onset. This interaction between genes and the environment can cause changes, like DNA methylation, which can turn genes on or off and contribute to the disorder’s progression.
Schizophrenia symptoms are categorized into three main groups:
Transcranial Magnetic Stimulation (TMS) is a groundbreaking treatment for managing schizophrenia symptoms. It addresses neural disruptions by using magnetic pulses to stimulate targeted brain areas.
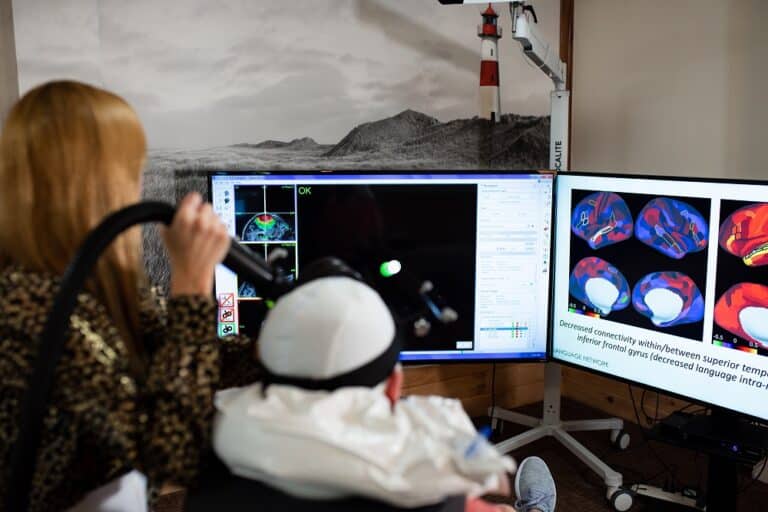
At Neurotherapeutix, we go beyond standard TMS by incorporating advanced resting-state functional connectivity MRI (rsFC MRI) to create personalized, effective treatment plans for individuals with schizophrenia.
Functional MRI (fMRI) plays a pivotal role in advancing the treatment of schizophrenia at Neurotherapeutix. This cutting-edge imaging technique provides real-time insights into brain activity, enabling a deeper understanding of how schizophrenia disrupts neural networks.

fMRI measures changes in blood flow within the brain, revealing patterns of activity and connectivity unique to each individual. In schizophrenia, fMRI allows us to:
Schizophrenia affects brain function through disruptions in connectivity between critical networks. These disruptions—referred to as dysconnectivity—can lead to a wide range of symptoms, including hallucinations, delusions, and cognitive impairments. TMS therapy works by restoring connectivity and function in these impaired networks.
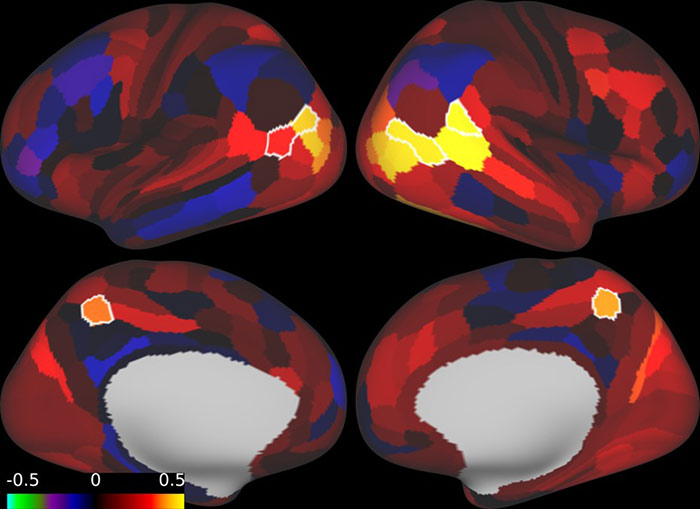
At Neurotherapeutix, we use rsFC MRI to map brain networks, providing insights into functional connectivity abnormalities specific to schizophrenia. Key findings include:
We pinpoint areas for TMS treatment by identifying these patterns,
TMS therapy offers a range of benefits for individuals with schizophrenia, providing a safe, effective, and personalized approach to symptom management.
By targeting the specific neural networks affected by the condition, TMS helps address positive and negative symptoms, ultimately improving patients’ quality of life.
Whether you are struggling with persistent symptoms or seeking an alternative to traditional treatments, fMRI TMS therapy provides an innovative, patient-centered approach to schizophrenia care.
fMRI therapy for schizophrenia involves three simple steps:
TMS therapy for schizophrenia at Neurotherapeutix offers an innovative solution to managing symptoms and improving quality of life. Contact us today to learn more about this transformative approach.
At Neurotherapeutix, we are committed to providing innovative, evidence-based care for individuals with schizophrenia. Using advanced technologies like fMRI TMS therapy, we offer a personalized, noninvasive approach that targets the root causes of symptoms, helping patients regain control of their lives.
If you or a loved one is seeking effective schizophrenia treatment in NYC, contact us today or request an appointment online. Together, we can create a path toward better mental health and lasting relief.

Yes, TMS therapy has shown effectiveness in managing symptoms of schizophrenia. It helps reduce positive symptoms like hallucinations and delusions and improves negative symptoms such as apathy and lack of motivation. Clinical studies also support its use for enhancing cognitive functions, making it a valuable tool in schizophrenia treatment.

While TMS is FDA-approved for several mental health conditions, including depression, ongoing research highlights its effectiveness for schizophrenia symptoms.

TMS therapy is highly effective when tailored to individual brain connectivity patterns.

TMS therapy typically involves multiple sessions over several weeks. Many patients notice symptom improvements within the first few weeks of treatment, but the duration of therapy may vary based on individual needs.

Yes, TMS therapy is a non-invasive and well-tolerated treatment with minimal side effects. It does not require surgery or medication, making it a safe option for individuals seeking alternative schizophrenia treatments.

At Neurotherapeutix, we use fMRI to map brain activity and identify specific connectivity disruptions associated with schizophrenia symptoms. This advanced imaging allows us to tailor TMS therapy to target the precise areas of the brain that need treatment, maximizing effectiveness and minimizing unnecessary stimulation.
Call us at (917) 388-3090 or click to request a regular or telehealth appointment.
Neurotherapeutix
171 East 74th Street, Unit 1-1 New York, NY 10021

Neurotherapeutix is the leading clinic for functional imaging guided transcranial magnetic stimulation (TMS), a safe, innovative, and non-invasive methodology for treating a wide range of acute and chronic mental disorders and brain injuries. Our advanced fMRI technology allows us to map the brain for the… Learn More »
By: Neurotherapeutix NYC
Reviewed By: Alayar Kangarlu, Ph.D.
Published: April 11, 2022
Last Reviewed: March 5, 2026
QUICK INQUIRY
Contact us to get an estimate for your medical services requirements. You can fill in the form to specify your medical requirements or you can call us directly.